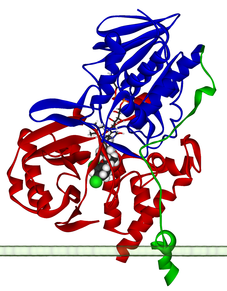
This web page was produced as an assignment for Genetics 564, an undergraduate course at UW‐Madison.
The MAOA protein
The MAOA protein is a mitochondrial enzyme in charge of catalyzing the oxidative deamination of biogenic and
xenobiotic amines, along with essential functions in the metabolism of neuroactive and vasoactive amines in the
central nervous system and peripheral tissues. Biogenic amines like 5-hydroxytryptamine (5-HT), norepinephrine
and epinephrine have higher prioritization for oxidation by the MAOA protein. [1,2]
xenobiotic amines, along with essential functions in the metabolism of neuroactive and vasoactive amines in the
central nervous system and peripheral tissues. Biogenic amines like 5-hydroxytryptamine (5-HT), norepinephrine
and epinephrine have higher prioritization for oxidation by the MAOA protein. [1,2]
|
Official name Cofactor Catalytic activity Isoelectric point Isoform 1 Molecular weight Accession number GI number Protein sequence Isoform 2 Molecular weight Accession number GI number Protein sequence |
: amine oxidase [flavin-containing] A : flavin adenine dinucleotide (FAD) : RCH2NHR' + H2O + O2 → RCHO + R'NH2 + H2O2 : 7.94 : 59682 dalton : NP_000231.1 : 4557735 : FASTA (527aa) : 44848 dalton : NP_001257387.1 : 395132504 : FASTA (394aa) |
Locations of MAOA proteins
MAOA protein is positioned at the outer mitochondrial membrane in neurons and astrocytes in the central nervous system (i.e. brain and spinal cord) to control both monoamine neurotransmitter availability for sequestration and the following extrasynaptic inactivation after release. [2] Outside the central nervous system, MAOA proteins are also found in the heart, liver, duodenum, blood vessels and kidney. [4]
Role of MAOA protein in serotonin catabolism
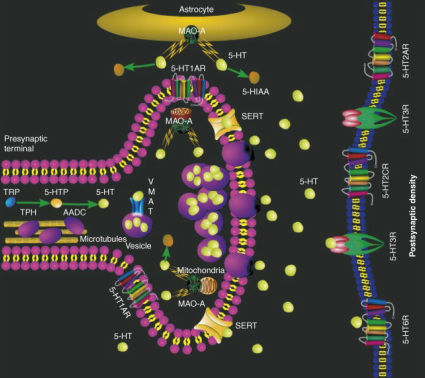
|
Serotonin (5-hydroxytryptamine; 5-HT) is hydroxylated to 5-hydroxytryptophan (5-HTP) from tryptophan (TRP) by tryptophan hydroxylase (TPH) and by the decarboxylation of the intermediate 5-HTP by amino acid decarboxylase (AADC). The vesicular monoamine transporter (VMAT) packages serotonin into synaptic vesicles. Serotonin can be degraded into 5-hydroxyindoleacetic acid (5-HIAA) presynaptically by mitochondrial MAOA, or extrasynaptically by glially-expressed MAOA. The clearing or synaptic serotonin can be carried out by either the serotonin transporter (SERT) or by binding to serotonin receptors residing on the presynaptic and postsynaptic membranes. [2]
Levels of expression
There are two transcript variants of MAOA in humans due to polymorphism in cDNA that affect the transcriptional activity of the gene promoter: Variant 1 is the high-activity form which produces more of the proteins (H-MAOA), whereas Variant 2 is the low-activity form which produces less of the proteins (L-MAOA). [5]
Transcript Variant 1 and Transcript Variant 2 give rise to Isoform 1 and Isoform 2 respectively. Isoform 1 consists of a longer sequence (527 amino acids) and is chosen as the 'canonical' sequence. Transcript Variant 2 contains an alternate exon in the 5' UTR and uses a more downstream start codon compared to the Transcript Variant 1. [6] Thus, amino acids at positions 1 to 133 in Isoform 1 are missing in Isoform 2, causing Isoform 2 to have a shorter sequence (394 amino acids), or specifically, a shorter N-terminus compared to Isoform 1. [4]
Low level or complete deficiency of MAOA has been shown to be associated with aggression. Studies simulated that the genotype of L-MAOA in combination with traumatic premature life events increases tendencies toward criminal aggression. [2] Complete MAOA deficiency caused by a missense mutation that yields a premature stop codon in the eighth exon of the MAOA gene is known to result in Brunner Syndrome, which is characterized by phenotypes like mild mental retardation, tendency to aggressive outbursts and violent impulsive behaviors. [8]
Transcript Variant 1 and Transcript Variant 2 give rise to Isoform 1 and Isoform 2 respectively. Isoform 1 consists of a longer sequence (527 amino acids) and is chosen as the 'canonical' sequence. Transcript Variant 2 contains an alternate exon in the 5' UTR and uses a more downstream start codon compared to the Transcript Variant 1. [6] Thus, amino acids at positions 1 to 133 in Isoform 1 are missing in Isoform 2, causing Isoform 2 to have a shorter sequence (394 amino acids), or specifically, a shorter N-terminus compared to Isoform 1. [4]
Low level or complete deficiency of MAOA has been shown to be associated with aggression. Studies simulated that the genotype of L-MAOA in combination with traumatic premature life events increases tendencies toward criminal aggression. [2] Complete MAOA deficiency caused by a missense mutation that yields a premature stop codon in the eighth exon of the MAOA gene is known to result in Brunner Syndrome, which is characterized by phenotypes like mild mental retardation, tendency to aggressive outbursts and violent impulsive behaviors. [8]
References:
- MAOA. National Center for Biotechnology Information. Retrieved Feb 2, 2014 from http://www.ncbi.nlm.nih.gov/gene/4128
- Buckholtz, J. W. & Meyer-Lindenberg, A. (2008). Maoa and the neurogenetic architecture of human aggression. Trends in Neurosciences, 31(3), 120-129.
- Protein image source: Wikipedia
- P21397 (AOFA_HUMAN). UniProtKB. Retrieved Feb 2, 2014.
- Sabol, S.Z., Hu, S., & Hamer, D. (1998). A functional polymorphism in the monoamine oxidase A gene promoter. Human Genetics,
- 103 (3), 273–9.
- MAOA monoamine oxidase A [Homo sapiens (human)]. Genetics Home Reference. Retrieved Feb 20, 2014.
- Brunner, H.G., et al. (1993). Abnormal behavior associated with a point mutation in the structural gene for monoamine oxidase A. Science, 262(5133), 578-580.