This web page was produced as an assignment for Genetics 564, an undergraduate course at UW‐Madison.
What are protein domains?
Domains are conserved structural and functional units of a protein. A protein consists of one to several domains; each domain forms a compact three-dimensional structure that can exist, fold and evolve independently. [1]
The MAOA protein domain: amino_oxidase
Human MAOA protein contains a single domain, namely the amino_oxidase domain which plays an important role in regulating the intracellular levels of amines via oxidation. [2] This domain contains two glycine-rich regions, both of which probably serves as a binding site for flavin adenine dinucleotide (FAD). The first region lies in the N-terminal
section whereas the second one is in the C-terminal section. [3]
Amino_oxidase belongs to the clan NADP_Rossmann, a class of redox enzymes with two domains: the catalytic domain confers substrate specificity and the precise reaction of the enzyme, whereas the Rossmann domain binds cofactor nicotinamide adenine dinucleotide (NAD+). The Rossmann domain consists of a central β-sheet surrounded
by around 5 α-helixes. The NAD+ binding site lies in the β-sheet. In some more distantly related Rossmann domains, NAD+ is replaced by FAD. [4]
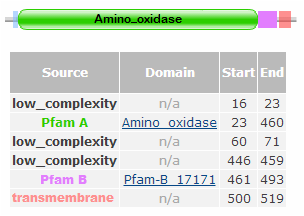
Identifiers of amino_oxidase are listed below.
section whereas the second one is in the C-terminal section. [3]
Amino_oxidase belongs to the clan NADP_Rossmann, a class of redox enzymes with two domains: the catalytic domain confers substrate specificity and the precise reaction of the enzyme, whereas the Rossmann domain binds cofactor nicotinamide adenine dinucleotide (NAD+). The Rossmann domain consists of a central β-sheet surrounded
by around 5 α-helixes. The NAD+ binding site lies in the β-sheet. In some more distantly related Rossmann domains, NAD+ is replaced by FAD. [4]
Identifiers of amino_oxidase are listed below.
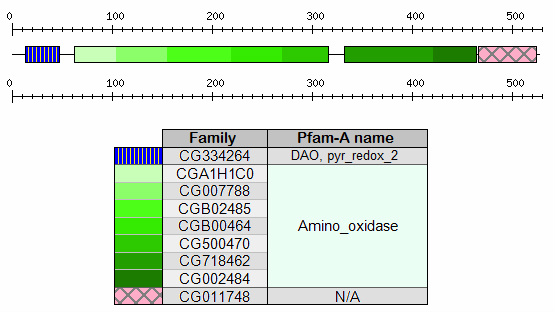
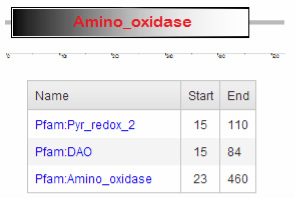
Figure 3. Human MAOA protein domains identified by ProDOM
As shown in Figure 2 and Figure 3, two signatures of less significance, DAO and pyr_redox_2 are also identified upstream of amino_oxidase domain. DAO stands for D-amino acid oxidase (a.k.a. FAD dependent oxidoreductase),
a flavoenzyme that catalyzes the oxidation of D-amino acids into their corresponding keto-acids. [5] Pyr_redox_2
stands for pyridine nucleotide disulphide oxidoreductase, a NAD-binding site within a larger FAD-binding domain. [6]
a flavoenzyme that catalyzes the oxidation of D-amino acids into their corresponding keto-acids. [5] Pyr_redox_2
stands for pyridine nucleotide disulphide oxidoreductase, a NAD-binding site within a larger FAD-binding domain. [6]
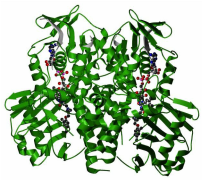
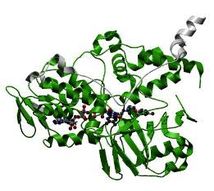
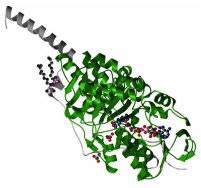
Structures of amino_oxidase domain
The parts in green are the amino_oxidase domains.
Domains in human MAOA isoforms
Differential transcriptional activities of the MAOA gene promoter result in two isoforms. Isoform 1 consists of a longer sequence (527 amino acids) and is chosen as the 'canonical' sequence for human MAOA protein. Amino acids at positions 1 to 133 in Isoform 1 are missing in Isoform 2, therefore Isoform 2 has a shorter sequence (394 amino acids) and a shorter N-terminus compared to Isoform 1, [7] meaning that Isoform 2 has a smaller or partial amino_oxidase
domain. Besides, DAO and pyr_redox_2 are not observed in Isoform 2 since they are both found within the range of Isoform 1 that is missing in Isoform 2. Thus, Isoform 2 might have a reduced functionality compared to Isoform 1.
domain. Besides, DAO and pyr_redox_2 are not observed in Isoform 2 since they are both found within the range of Isoform 1 that is missing in Isoform 2. Thus, Isoform 2 might have a reduced functionality compared to Isoform 1.
Domains in MAOA homologs
Mouse, dog, cow, and cat all have MAOA protein sequence of the same length as human's (Isoform 1), hence it is not surprising that the domain amino_oxidase was found at the exact same location in these homologs. Chimpanzee, monkey and chicken have MAOA protein sequence of shorter length compared to human, which explains why the amino_oxidase domain found in these three homologs are only partial. When aligned, it can be observed that the N-terminal section of human MAOA protein sequence is missing in chimpanzee, monkey and chicken's homologs, causing around one third of amino_oxidase domain near the N-terminus present in human MAOA protein to be missing in these three homologs as well. To study the functions of MAOA protein, mouse, dog, cow and cat might be a better choice of model organisms than chimpanzee and chicken, since the amino_oxidase domain is better conserved in the homologs of the former group.

Figure 5. Amino_oxidase domain in MAOA protein homologs identified by PFAM
References:
- What are protein domains? EMBL-EBI. Retrieved March 21, 2014.
- Family: Amino_oxidase (PF01593). Pfam. Retrieved March 22, 2014.
- PROSITE documentation PDOC00755. PROSITE. Retrieved March 22, 2014.
- Clan: NADP_Rossmann (CL0063). Pfam. Retrieved March 22, 2014.
- Family: DAO (PF01266). Pfam. Retrieved March 22, 2014.
- Family: Pyr_redox_2 (PF07992). Pfam. Retrieved March 22, 2014.
- P21397 (AOFA_HUMAN). UniProtKB. Retrieved Feb 2, 2014.
- MAOA monoamine oxidase A. National Center for Biotechnology Information. Retrieved Feb 20, 2014.