This web page was produced as an assignment for Genetics 564, an undergraduate course at UW‐Madison.
Aggression is a multi-dimensional concept, but it can be generally defined as any behavior or disposition towards others involving force, hostility or assault.[1] Aggression can be categorized into two general groups: retaliatory aggression which stems from negative emotions and instrumental aggression which is projected to achieve a goal without being provoked.[2] Real-life aggression involve mixed motives and interacting causes,[3] and persistent aggression often leads to criminality.
|
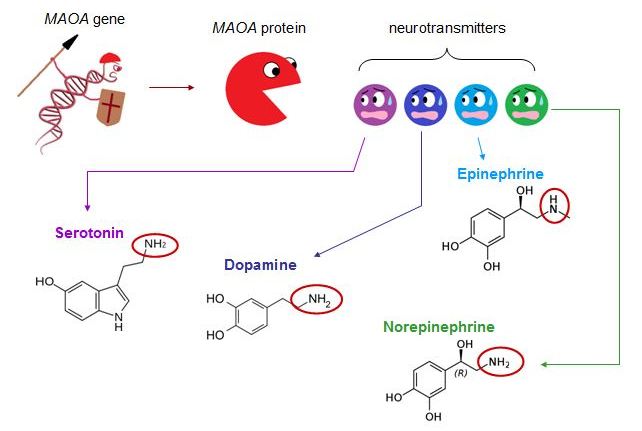
Monoamine oxidase A (MAOA) gene, nicknamed the “warrior gene”, has been discovered to be the most compelling candidate for a gene of susceptibility for aggression. The MAOA gene encodes for MAOA protein, an enzyme that deaminates several neurotransmitters— norepinephrine, epinephrine, serotonin and dopamine.[4] |
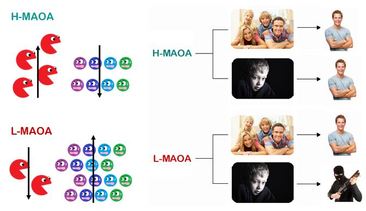
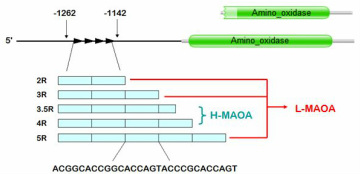
L-MAOA produces less MAOA proteins and causes the mentioned neurotransmitters to build up in excess, thereby leading to increased propensity towards aggression.[5-7] Clinical imaging work has demonstrated that people with the low-activity form of the MAOA gene (L-MAOA) display greater reactivity in the amygdala and lower activity in the regulatory prefrontal areas during affective arousal, suggesting that the emotional and cognitive channels link L-MAOA to impulsive forms of aggression.[8] Further studies simulated the threat of L-MAOA in combination with traumatic premature life events increase both psychiatric patients and healthy adults’ tendencies toward violence.[9] It has been proposed that when subjected to abusive childhood, an L-MAOA individual has a significantly heightened chance of generating Anti-Social Personality Disorder (ASPD).[10] ASPD is a mental health condition in which a person has a long-term pattern of manipulating, exploiting, or violating the rights of others, and is often criminal.[11]
By taking an approach informed by the modern scientific literature, we may be able to improve their lives for the better and benefit the society by preventing the occurrence of aggressive behaviors, which in turn lower crime rate. Targeted intervention and early administration would be helpful to prevent manifestation of ASPD and to keep psychopathy under control. Alongside with psychological therapy, it might be more effective to also give medical treatment to the affected individuals. However, supplementation of drugs to suppress aggression has not been an option, because the interactions of human MAOA with other proteins are not fully understood. Direct supplementation of MAOA is impractical because mass production of MAOA is almost impossible. Extraction and production of MAOA proteins involve difficult procedures that require long processing time and high cost.
The alternative is to look for drugs that can upregulate MAOA, is orally biodegradable, potent and active in vivo.
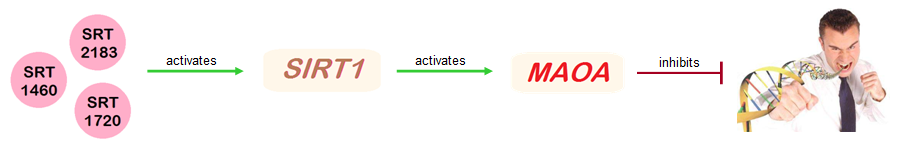
The SIRT1 gene has been shown to activate the transcription of the MAOA gene in mice.[12] Given this, I hypothesize that upregulation of SIRT1 will increase MAOA levels and suppress aggression. It was discovered that three novel small-molecule activators of SIRT1, namely SRT2183, SRT1460 and SRT1720, are able to increase the expression of SIRT1 in rats and humans.[13] Most importantly, these small-molecule activators are potent, orally bioavailable and active in vivo.[14]
The SIRT1 gene has been shown to activate the transcription of the MAOA gene in mice.[12] Given this, I hypothesize that upregulation of SIRT1 will increase MAOA levels and suppress aggression. It was discovered that three novel small-molecule activators of SIRT1, namely SRT2183, SRT1460 and SRT1720, are able to increase the expression of SIRT1 in rats and humans.[13] Most importantly, these small-molecule activators are potent, orally bioavailable and active in vivo.[14]
|
I will use rhesus monkeys (Macaca mulatta) to test the hypothesis that small-molecule activators of SIRT1 can increase MAOA level in the brain and suppress aggression. Primates make good model organisms for a behavioral trait like aggression because they have cognitive and emotional functioning that is similar to human. I choose rhesus monkey over chimpanzee because the majority of chimpanzee carry a single repeat of the VNTR sequence similar to that in human, whereas 40% of Rhesus monkey carry the L-MAOA allele. [15] |
My primary goal is to determine whether the upregulation of SIRT1 (through supplementation of SRT2183, SRT1460
and SRT1720) increases MAOA levels and thus, reduces aggressive behaviors in L-MAOA rhesus monkeys. I will then examine the mechanisms of the interactions between SIRT1 and MAOA in rhesus macaques. The objective of this study is to analyze the feasibility of SRT2183, SRT1460 and SRT1720 as potential drugs to upregulate MAOA
and suppress aggression in L-MAOA human.
and SRT1720) increases MAOA levels and thus, reduces aggressive behaviors in L-MAOA rhesus monkeys. I will then examine the mechanisms of the interactions between SIRT1 and MAOA in rhesus macaques. The objective of this study is to analyze the feasibility of SRT2183, SRT1460 and SRT1720 as potential drugs to upregulate MAOA
and suppress aggression in L-MAOA human.
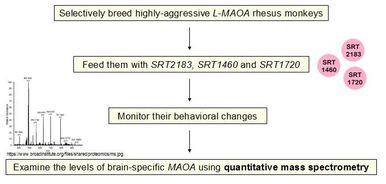
My Specific Aim 1 is to determine whether
SRT2183, SRT1460 and SRT1720 can increase MAOA levels in the brain and reduce aggression levels in L-MAOA rhesus monkeys.
I will selectively breed highly-aggressive L-MAOA
rhesus monkeys, feed them with SRT2183,
SRT1460 and SRT1720, access their behavioral
changes, and observe the levels of MAOA
proteins in their brain samples using quantitative mass spectrometry. My hypothesis is that supplementation of SRT2183, SRT1460 and SRT1720 will upregulate MAOA levels in brain and therefore prevent manifestation of aggressive behaviors in rhesus monkeys. Future research will be to repeat the study in human to determine if SRT2183, SRT1460 and SRT1720 can be drugs to suppress aggression in MAOA-deficient individuals.
SRT2183, SRT1460 and SRT1720 can increase MAOA levels in the brain and reduce aggression levels in L-MAOA rhesus monkeys.
I will selectively breed highly-aggressive L-MAOA
rhesus monkeys, feed them with SRT2183,
SRT1460 and SRT1720, access their behavioral
changes, and observe the levels of MAOA
proteins in their brain samples using quantitative mass spectrometry. My hypothesis is that supplementation of SRT2183, SRT1460 and SRT1720 will upregulate MAOA levels in brain and therefore prevent manifestation of aggressive behaviors in rhesus monkeys. Future research will be to repeat the study in human to determine if SRT2183, SRT1460 and SRT1720 can be drugs to suppress aggression in MAOA-deficient individuals.
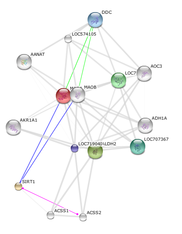
My Specific Aim 2 is to elucidate the mechanism of how SIRT1 expression affect brain-specific MAOA expression in rhesus monkeys. I will extract brain samples of both L-MAOA and H-MAOA rhesus monkeys, then use TAP-tags in conjunction with Y2H system to examine mechanism of how SIRT1 expression affect MAOA expression. My hypothesis is that SIRT1 protein is a key transcriptional upregulator of MAOA gene. The diagram on the right is a predicted network of protein-protein interaction generated using STRING database. The red molecule represents MAOA protein, and the yellow molecule represents SIRT1 protein. I will be paying attention to the proteins that act as transcription factors of MAOA. A better understanding of how MAOA and SIRT1 interact with each other and with their protein partners will provide a reference for the homologous pathways in human.
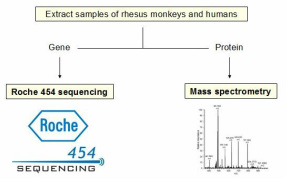
My Specific Aim 3 is to study the homology of SIRT1 and MAOA variants (both genes and proteins) between rhesus monkeys and humans. I will use Roche454 sequencing to determine the variants of SIRT1 and MAOA genes in rhesus monkeys and humans, then compare their homology. I choose Roche454 over Illumina mainly because Roche454 allows for de novo assembly, whereas Illumina requires a reference sequence. To obtain more accurate results of all the variant sequences, especially the alignments at the regions containing of VNTR (Variable Number Tandem Repeat), de novo assembly can probably do a better job as aligning without a reference sequence tend to be less biased. For homology analysis of SIRT1 and MAOA proteins in Rhesus monkeys and humans, I will use mass spectrometry. My hypothesis is that SIRT1 and MAOA are both very well-conserved in rhesus monkeys and humans, with high similarity in the patterns of polymorphism. Future research will be focusing on post-transcriptional modifications and phosphorylation sites of the genes, as well as post-translational tertiary structure modifications (folding) of the proteins.
Overall, future directions will be to duplicate the study in human to gain a better understanding between SIRT1, MAOA and aggression in human, so that medical treatment can be made possible to better the lives of the affected L-MAOA individuals. Further research on the mechanism behind MAOA and aggression is hoped to provide a different view of criminality in the fields of psychology, psychiatry, social work and judiciary system.
| Presentation slides | |
| File Size: | 2283 kb |
| File Type: | |
References:
- Aggression. Merriam-Webster Online. Retrieved Feb 2, 2014.
- Berkowitz, L. (1993). Aggression: Its causes, consequences, and control. New York, NY: McGraw-Hill.
- Bushman, B.J.; Anderson, C.A. (2001). Is it time to pull the plug on the hostile versus instrumental aggression dichotomy? Psychological Review 108(1): 273–279.
- Guo, G., Ou, X., Roettger, M., & Shih, J. C. (2008). The VNTR 2 repeat in MAOA and delinquent behavior in adolescence and young adulthood: associations and MAOA promoter activity. European Journal of Human Genetics, 16(5), 626-634.
- Grimsby, J., Chen, K., Wang, L.J., Lan, N.C., & Shih, J.C. (1991) Human monoamine oxidase A and B genes exhibit identical exon-intron organization. Proceedings of the National Academy of Sciences of the United States of America, 88(9):3637–3641.
- Sabol, S.Z., Hu, S., & Hamer, D. (1998). A functional polymorphism in the monoamine oxidase A gene promoter. Human Genetics, 103(3), 273–9.
- Beaver, K. M., Wright, J. P., Boutwell, B. B., Barnes, J., Delisi, M., & Vaughn, M. G. (2013). Exploring the association between the 2-repeat allele of the MAOA gene promoter polymorphism and psychopathic personality traits, arrests, incarceration, and lifetime antisocial behavior. Personality and Individual Differences, 54(2), 164-168.
- Meyer-Lindenberg, A., et al. (2006). Neural mechanisms of geneticrisk for impulsivity and violence in humans. Proceedings of the National Academy of Sciences of the United States of America, 103(16), 6269-6274.
- Frazzetto, G., Di Lorenzo, G., Carola, V., Proietti, L., Sokolowska, E., et al. (2007) Early Trauma and Increased Risk for Physical Aggression during Adulthood: The Moderating Role of MAOA Genotype. PLoS ONE 2(5): e486.
- Caspi, A., et al. (2002) Role of genotype in the cycle of violence in maltreated children. Science, 297(5582), 851– 854.
- Antisocial Personality Disorder. PudMed Health. Retrieved March 3, 2014.
- Libert, S., Rubio, J. P., Asara, J., Cohen, D., Das, A., Bell, E., et al. (2011). SIRT1 Activates MAO-A in the Brain to Mediate Anxiety and Exploratory Drive. Cell, 147(7), 1459-1472.
- Milne, J. C., Lynch, A. V., Olefsky, J. M., Jirousek, M. R., Elliott, P. J., Nunes, J. J., et al. (2007). Small molecule activators of SIRT1 as therapeutics for the treatment of type 2 diabetes. Nature, 450(7170), 712-716.
- Milne, J. C., & Denu, J. M. (2008). The Sirtuin family: therapeutic targets to treat diseases of aging. Current Opinion in Chemical Biology, 12(1), 11-17.
- Wendland, J. R., Hampe, M., Newman, T. K., Syagailo, Y., Meyer, J., Schempp, W., Timme, A., Suomi, S. J. and Lesch, K. P. (2006), Structural variation of the monoamine oxidase A gene promoter repeat polymorphism in nonhuman primates. Genes, Brain and Behavior, 5: 40–45.